METTLER TOLEDO (Switzerland) - In pharmaceutical manufacturing, inaccurately measured ingredients can lead to bad batches and waste. If not caught in the manufacturing process, defective products could end up in the hands of consumers, potentially causing physical harm. METTLER TOLEDO has recently published a webinar that explains the importance of weighing processes to ensure consistent final product quality.
Carefully selecting a weighing solution to match specific process requirements is critical in pharmaceutical manufacturing because weighing processes can contribute significantly to consistent final product quality.
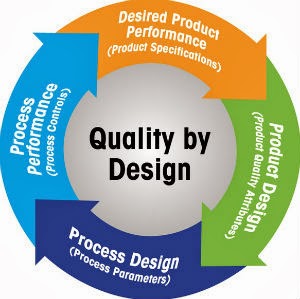
When developing processes based on the Quality-by-Design (QbD) concept, steps should be taken to ensure quality is built in.
QbD incorporates the entire development and production lifecycle, including specification of the desired product performance, product design, process design and process performance to ensure quality in every process step. It is a scientific, risk-based approach to identify process errors early so that proactive steps may be taken to ensure safety and efficacy of pharmaceuticals.
To explain the potential influence of QbD in weighing processes and product quality, METTLER TOLEDO experts have recently published a new webinar, "QbD Ensuring Consistent Weighing Quality in Pharmaceutical Manufacturing." This webinar provides an overview on various aspects of weighing quality within pharmaceutical manufacturing and examines the relevance of weighing processes for QbD frameworks. It is an educational tool for anyone involved with quality control in pharmaceutical manufacturing.
To download the free white paper click here.
Source Link
Other Articles from METTLER TOLEDO (Switzerland)
© METTLER TOLEDO (Switzerland) / International Weighing Review
This website uses cookies. By browsing our website you accept the use of cookies. For more information read our Terms of Use.
New Webinar Explains Relevance of Weighing Processes in Quality by Design Concepts
- By METTLER TOLEDO
- Posted on Dec 10, 2013
For more information about this article from METTLER TOLEDO click here.
Other articles from METTLER TOLEDO.
Interesting Links:
List all your Scales and other Weighing Products (new and used) on our Marketplace !
Visit AutomationInside.com for Automation and Technology Related News!
Are you interested to include your Link here, visible on all WeighingReview.com articles and marketplace product pages? Contact us
List all your Scales and other Weighing Products (new and used) on our Marketplace !
Visit AutomationInside.com for Automation and Technology Related News!
Are you interested to include your Link here, visible on all WeighingReview.com articles and marketplace product pages? Contact us
© METTLER TOLEDO / International Weighing Review
Previous article
New Approvals for Zemic Europe Stainless Steel IP69K Single Point Load Cell
Next article
Eilon Engineering Load Monitoring Useful Across a Variety of Industries
Popular News
Weighing Review Newsletter
Popular Tags
Login





















Interested? Submit your enquiry using the form below:
Only available for registered users. Sign In to your account or register here.