SG Systems (USA) - Once again, SG Systems’ is leading the way with their formula control & ingredient traceability solution; this time with Batesville, AR based Lifeplus International.
Lifeplus President Bob Lemon, R.Ph, began his professional career as a pharmacist, providing prescriptions to thousands through a number of successful pharmacies based in the US.
Seeking to better service his customers, and with the foresight to recognize that nutrition would begin to play an increasingly prominent role in modern medicine, it became his mission to make high quality nutritional supplements available directly to the consumer. He went on to purchase a respected nutritional supplements manufacturer based in the US, laying the foundations for Lifeplus, which was founded in 1992.
When he later met with Dr Dwight McKee, a practitioner with a shared vision for the role of nutrition in healthcare, he recognized an opportunity to create a partnership that would eventually lead to an extensive range of nutritional products based on the latest science, practitioner experience and the highest standards – available to all.
Today, Lifeplus has a worldwide distribution and an enviable reputation for high quality Dietary Supplements, Vitamins & Minerals, Nutritional Shakes, Weight Management, and Antioxidants plus many more inspiring innovations. All of which are lab controlled and manufactured to exacting tolerances in their state of the art facility closely following FDA mandated Good Manufacturing Practices.
For these reasons, Lifeplus decided to invest in the Vantage Formula Control and Lot Traceability Solution from SG Systems LLC. Lifeplus needed to take their manual batching process with paper-based lot verification to their next level and bridge the gap between their Fourth Shift ERP System and the shop floor weighing operations. Fourth Shift typically manages a company’s business, including the Bills of Materials (BOM), Production Schedule and Inventory Traceability.
Companies usually print the Production Schedule and BOMS on a sheet of paper and hand it to the production team to complete the weighing and batching processes, and then return the completed paperwork to the production supervisors to enter the inventory used and batches made into the ERP System. This process is now well known to be problematic. Operator errors in the weighing process and inaccuracies in lot control are usually not reflected in the paper trail – this can cause problems with accounting, inventory and purchasing in addition to the quality of the product from batch to batch.
Traceability Solution – Vantage:
It all starts from a download from Fourth Shift. Once a manufacturing order has been created in Fourth Shift, a file is transferred to the Stevens Integration Services Software containing the Production Plan (number of batches, required batch size and production locations) and the BOM containing the recipe steps, weighing sequence and target weights. The available inventory is made available for the system to consume from (lot numbers, stock codes and quantities).
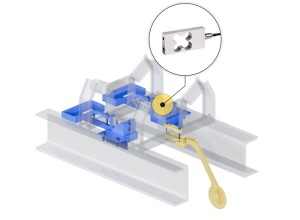
The Production Plan is then available at the 2 operator workstations. Each station consists of a Vantage Traceability Touch Screen User Interface, Label Printer and Barcode Scanner. The steelwork holds 2 scales at each workstation. The main scale is a heavy duty unit designed for the heavier items and the small weights are weighed on a high resolution balance.
The Vantage Traceability Touch Screen User Interface allows the Production Plan (transferred from Fourth Shift) to be displayed using easy to follow screens. This eliminates paper traceability schedules and prevents operators making incorrect or out of sequence formulations. After selecting the required formula, the operator follows simple screens which prompt for lot number traceability scans. A speedometer display ensures the weighed items are portioned correctly and within tight, management set tolerances. The operator cannot weigh up too much or too little as the system will not accept out of tolerance weighing. This has a big impact of batch consistency and overall product quality.
‘Mistake Free’ Paperless Lot Traceability:
The FDA requires each batch to have a hard copy report detailing which operator has weighed up the respective steps, traceability detailing the lot number and scaled amount. The Recipe Traceability Batch Report allows this visibility, plus a section for three signatures to ensure complete acceptance and sign off for the batch traceability. The SG Systems Traceability Software has automated this process to ensure fact and accurate batch sign off.
To provide paperless lot traceability, the Vantage System creates inventory usage files in real time (after each batch is complete) which are in turn used to deduct traceability inventory from Fourth Shift. This improves inventory traceability visibility, reduces man hours spend on data entry and saves time double handling data. In addition, the Vantage System provides actual usage based on the scaling weights, instead of theoretical usage based on target weights. This allows accurate yields to be calculated which provides visibility for supplier inventory accuracy.
George Dust, Lifeplus General Manager comments “for diligence purposes, we conducted a thorough analysis of the systems available. SG Systems has a history of Recipe Formulation & Traceability Systems with installations in many countries over many years. For our business, it was important that Fourth Shift remained the master database. SG Systems was able to demonstrate a clear, safe and practical method for handling our data. In addition, SG Systems is a company that is flexible enough to customize their hardware to meet the needs of a demanding, high productivity, production led operation like ours. Now that we have the SG Systems Vantage Traceability installed, batches are achieved with much greater accuracy and traceability!”
Video:
This website uses cookies. By browsing our website you accept the use of cookies. For more information read our Terms of Use.
Formula Control Scales with ERP Interface from SG Systems at Dietary Supplements Company
- By SG Systems LLC
- Posted on May 15, 2014

For more information about this article from SG Systems LLC click here.
Other articles from SG Systems LLC.
Interesting Links:
List all your Scales and other Weighing Products (new and used) on our Marketplace !
Visit AutomationInside.com for Automation and Technology Related News!
Are you interested to include your Link here, visible on all WeighingReview.com articles and marketplace product pages? Contact us
List all your Scales and other Weighing Products (new and used) on our Marketplace !
Visit AutomationInside.com for Automation and Technology Related News!
Are you interested to include your Link here, visible on all WeighingReview.com articles and marketplace product pages? Contact us
© SG Systems LLC / International Weighing Review
Previous article
Ishida Europe receives Queen's Award for Enterprise
Next article
100% Rockwell Automation / Allen Bradley Checkweighers Enter the In-Motion Scale Market
Popular News
Weighing Review Newsletter
Popular Tags
Login












Interested? Submit your enquiry using the form below:
Only available for registered users. Sign In to your account or register here.